|
Our overarching goal is to uncover the molecular and cellular processes that regulate memory formation and persistence.
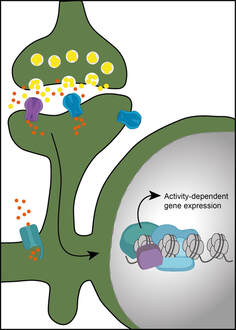
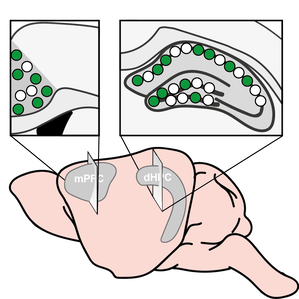
A key first step is memory formation is the activation of transcription triggered by learning. We focus on the processes that couple neuronal activity at the synapse with transcription activation in the nucleus. Epigenetic mechanisms are particularly important in the control of gene transcription. They can regulate gene accessibility via modulation of transcription factor binding and the structure of chromatin. We are interested in investigating whether epigenetic readers and writers regulate activity-dependent gene transcription and whether epigenetic factors are themselves regulated by neuronal activity. By uncovering these key players and mechanisms, we hope to shed light on the critical initial steps of memory formation. Key publications: Brito et al., JN, 2020; Brito et al., MB, 2020; Gulmez Karaca et al., IJMS, 2019; Gulmez Karaca et al., NLM, 2018; Oliveira, LM, 2016; Oliveira et al., NN, 2012. Memory representations or engrams are stored in neurons, also known as engram cells, that are activated during learning and reactivated by the retrieval of that memory. Upon the activation of engram cells during learning, a myriad of processes must take place that ensures that these cells are later reactivated to reconstruct aspects of the initial experience. We have currently virtually no understanding of the molecular mechanisms that regulate the persistence of memory and engram stability. The initial learning-triggered transcriptional response is mostly identical, independently of whether an experience will be remembered for short (days – recent memory) or long (weeks, month or a lifetime – remote memory) periods of time. Thus, additional mechanisms must occur that ensure the longevity of some memories. We aim at uncovering these processes through an interdisciplinary approach that leverages behavioral testing, engram tools, epigenetic and transcriptional analysis, as well as morphological and electrophysiological approaches. Key publications: Gulmez Karaca et al., BRB, 2021; Gulmez Karaca et al., NComms, 2020; Oliveira, LM, 2016. Ultimately, we are applying our gained knowledge of the molecular and cellular mechanisms of memory formation and maintenance to interrogate other forms of behavioral (mal)adaptations and pathological conditions. Namely, the mechanisms underlying age-dependent cognitive decline as well as persistent plasticity changes that drive a transition to maladaptive behavior for instance during the establishment of addictive states or aversive life experiences. Key publications: Gulmez Karaca et al., NA, 2021; Brito et al., JN, 2020; Oliveira et al., MP, 2019; Cannella et al., JN, 2018; Oliveira et al., MP, 2016; Oliveira et al., NN, 2012. |